This is followed by anhydrides and esters. Then acidify salt with diluted hydrochloric acid. Shake both tubes, observe, and record the results. To one tube add 3 mL of cold water, to the other add 3 mL of 10 sodium hydroxide solution. In addition, certain carboxylic acids can be oxidised.

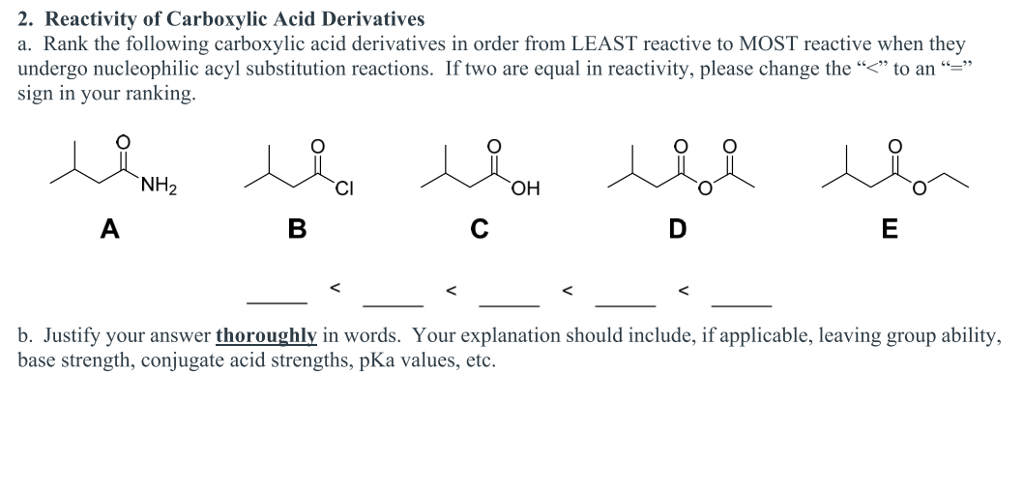
Carboxylic acids can also be reduced, decarboxylated and turned into acid derivatives. Carboxylic acids react with alcohols in the presence of a strong, concentrated acid catalyst to produce esters. (a) Salt Formation In each of two test tubes, place 0 g of benzoic acid. Carboxylic acids react with bases to form carboxylate salts. Presence of electronegative chloride on the carbonyl carbon makes it reactive towards nucleophilic substitution reaction. Part B: Tests of Carboxylic Acids, their Salts and Derivatives. The general formula of ester is R C O O R A c i d h a l i d e s > A c i d a n h y d r i d e s > E s t e r > A m i d e The ester is another derivative of carboxylic acid. The reaction is known as esterification reaction. Get detailed, expert explanations on relative reactivity of carboxylic acid derivatives that can improve your comprehension and help with homework.

The basic hydrolysis of an ester is also known as saponification and produces a water soluble. Furthermore, the ethoxide ion which is lost from the molecule is a stronger base than water and undergoes proto-nation (Fig. The acid chlorides react with alcohols to form esters. Learn all about relative reactivity of carboxylic acid derivatives. The carboxylic acid which is formed reacts with sodium hydroxide to form a water soluble carboxylate ion (Fig. Chemical structure contains a carbonyl function (-CO) and an hydroxyl group (OH), these groups interact easily with polar compounds, forming bridges of H.
